
At present, only a few companies in the world extract beryllium oxide from ores, such as China (such as Shuikoushan No. 6 Plant), Brash Wellman Company in the United States, and Urbinsky Metallurgical Plant in Kazakhstan. , the main production methods are sulfuric acid method and fluorination method
The Sulfuric Acid Method For Berylium
The sulfuric acid method is still one of the widely used methods in the production of modern beryllium hydroxide and beryllium oxide. The acid-soluble metal enters the solution phase, and is preliminarily separated from gangue minerals such as silicon, and then the beryllium-containing solution is purified to remove impurities, and finally a qualified beryllium oxide (or beryllium hydroxide) product is obtained.
As early as the 1940s, the German Degussa company used the sulfuric acid method (ie the Degussa process) to produce beryllium hydroxide. Subsequently, the American Brash Beryllium Company improved the process (ie, the Brush process). In 1969, the American Brash-Wellman Company built a combined extraction technology (ie, acid leaching-extraction process) to treat low-grade silicon beryllium. Stone and Beryl Factory. So far, the sulfuric acid method has been continuously improved and perfected.
Degussa Craft
The Degussa process is suitable for processing beryl minerals with high beryllium content. Since beryl cannot be directly decomposed by sulfuric acid, an alkali flux must be added or its crystal form or structure must be changed by heat treatment to increase the reactivity before acid hydrolysis. The reaction is:
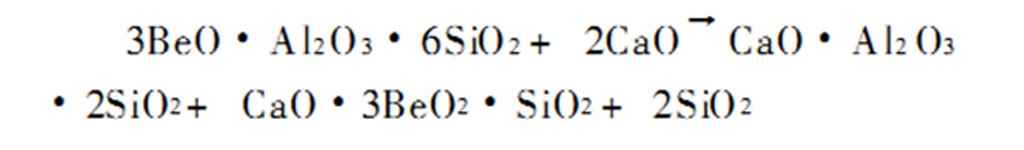
The added flux can be an alkaline oxide such as soda ash, lime, etc., or a chloride such as calcium chloride, sodium chloride, etc. Among them, lime has the advantages of price and environmental protection. During roasting, the proportion of ingredients (m lime/m beryl) is usually controlled to 1~3, and the roasting temperature is generally 1400~1500℃.
Brush Craft
The Brush process eliminates the step of adding flux, and directly heats beryl in an electric arc furnace to 1700°C to melt, and then pours it into high-speed flowing cold water to obtain granular beryllium glass, which is then heated to 900°C in a gas furnace to precipitate beryllium oxide. After crushing, it is mixed with 93% sulfuric acid to form a slurry, and the slurry is acidified at 250~300℃, and the leaching rate of beryllium in the ore can reach 93%~95%. The main reactions of the process are as follows:
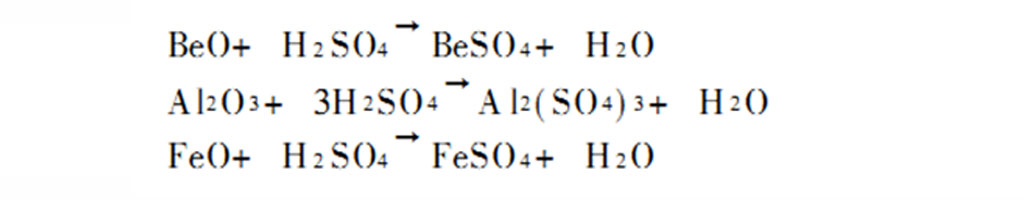
Acid Leaching-Extraction Process
In the 1960s, the U.S. Bureau of Mines used an acid leaching-extraction process to treat beryllium from Utah and beryl from Kings Mountain, North Carolina. In 1969, the American Brash-Wellman Company established a plant in Delta, Utah, to process low-grade beryllium by sulfuric acid-extraction process.
The beryllium stone is first smashed and roasted, and then roasted and broken (sprayed with water to avoid dust flying) and then wet-milled to <0.07mm in a ball mill with a classifier. wash water. After wet grinding, add 10% sulfuric acid to the pulp, stir and pickle it for 24 hours under the conditions of a liquid-solid ratio of 3 and a temperature of 65 °C, and then countercurrent sedimentation in a countercurrent pouring washing thickener to discard the slurry, and the obtained leachate contains beryllium. 0.4~0.7g/L, aluminum 4~7g/L, pH value 0.5~1.0. Eight-stage countercurrent extraction was carried out with bis(2-ethylhexyl)phosphonate (D2EHPA)-ethanol-kerosene as the organic phase, and beryllium and a small amount of aluminum and iron were discarded into the organic phase raffinate. The reaction formula of this extraction process is:
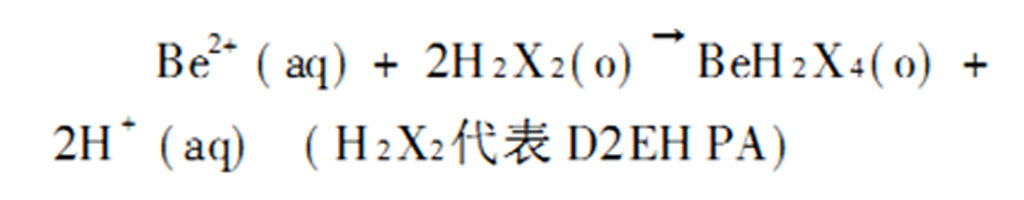
The obtained loaded organic phase is back-extracted with ammonium carbonate solution, and beryllium enters the water phase to form beryllium ammonium carbonate, and iron and aluminum also enter the water phase. The reaction formula of the back-extraction process is:
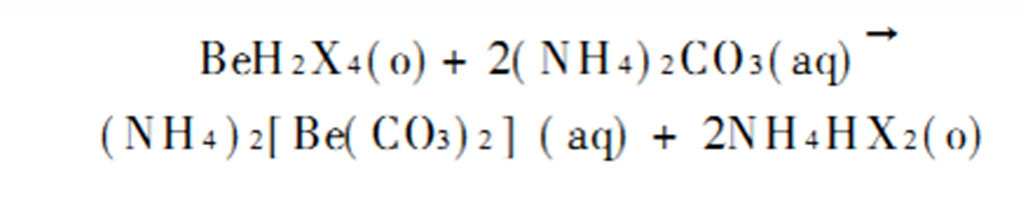
The organic phase after stripping is acidified and regenerated by sulfuric acid and then returned to the extraction process, and the stripping solution is heated to 70°C to separate iron and aluminum by hydrolysis and precipitation; then the solution after removing iron and aluminum is heated to 95°C, and EDTA is added. The complexing agent is used to hydrolyze the beryllium ammonium carbonate solution to obtain the basic beryllium carbonate precipitation. The filtered uranium-containing filtrate is used to recover uranium, and the filter cake is slurried with deionized water and then heated to 165 ° C with steam for hydrolysis. The main reaction is as follows ;
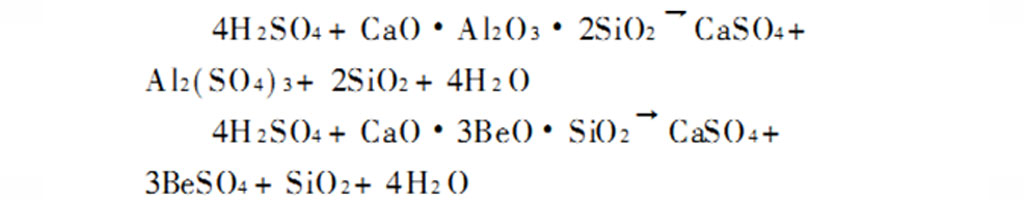
The basic beryllium carbonate precipitate and the beryllium-containing filtrate are obtained by hydrolysis, and the filtrate is added with alkali to precipitate beryllium hydroxide, which is used together with basic beryllium carbonate as a product. The acid leaching-extraction process has the following characteristics: both the organic phase and the back-extraction precipitation can be recycled, and the efficiency is high; except for the leaching residue, the discharged pollutants are only raffinate and pickling waste liquid, which are small in quantity and easy to handle; The stripping process is easy to achieve continuous and automatic; it can process ores with high impurity lithium and fluorine content and obtain high-quality beryllium oxide products.
Fluorination
The beryllium metal contains a lot of voids, and the microscopic study shows that it is a coarse grain structure. Even after processing, the grain size is larger than 100 μm. As a result, the strength performance of beryllium ingots is not high. Therefore, the manufacture of beryllium parts can only use Powder metallurgy, that is, the beryllium ingot is first turned into chips, then made into powder, and finally united and formed.
The chemical composition, physical properties and technological properties of beryllium powder have a significant impact on its consolidation process and the properties of the final product. The old powdering process can no longer meet the requirements of a new generation of high-strength and high-ductility beryllium materials. The progress of beryllium industry is closely linked with the development of beryllium powder metallurgy technology. The new beryllium powder preparation method that has been researched and applied in the past two decades is impact grinding, also known as cold flow impact, which is a new powder production process developed by using the cold brittleness of metal. It uses high-speed, high-pressure gas. Bombarding the beryllium target in the crushing chamber with the coarser particles through the nozzle, the pressure immediately drops from high pressure to atmospheric pressure. Adiabatic expansion occurs, which reduces the temperature of the beryllium target and the crushing chamber to room temperature or even below zero. At this time, the cooled particles are crushed by impact. Compared with the traditional disc milling and ball milling, this method has the advantages of high powder purity, uniform particle shape, large internal stress of the powder, high efficiency, low cost, and good protection.
In addition, the production of beryllium powder by gas atomization can also produce regular spherical particle powder, which can significantly reduce or eliminate the anisotropy of the briquette. The geometrically precise consolidation and the generally polycrystalline microstructure of the atomized beryllium powder are particularly important for the subsequent hot isostatic pressing process.
Pressure forming is the second largest process in beryllium powder metallurgy. In the past, vacuum hot pressing was mostly used, and in the 1980s, vacuum hot pressing beryllium materials were further developed. By changing the chemical composition, particle size distribution and processing temperature, a variety of plated products with different properties and a wide range of uses have been produced. A new technology that has developed rapidly in the past two decades is hot isostatic pressing technology. The performance of its products is far superior to that of vacuum hot pressing, especially to produce special-shaped beryllium parts with extremely complex structures and high-strength ductile beryllium The efficiency is greatly improved, and it is a relatively economical and promising process.